Smart Growth for
Private Business
Growth is CHANGE.
Growth requires more
people, processes and controls.
As a business grows, the
structure (organizational chart) has to change. It becomes more complex.
As a business grows, it needs better finance,
HR and technology tools
The 5 “P´s"
- PLANNING
- PRIORITZATION
- PACE
- PROCESSES
- PEOPLE
While strategic focus (Planning and Prioritization) and
operational excellence (Pace and Processes) are necessary
for growth, they are not sufficient.
Growth also requires the right kind of People, culture and leadership.
Growth requires the
hiring, training and retaining of high performance employees, and the building
of a high performance management team.
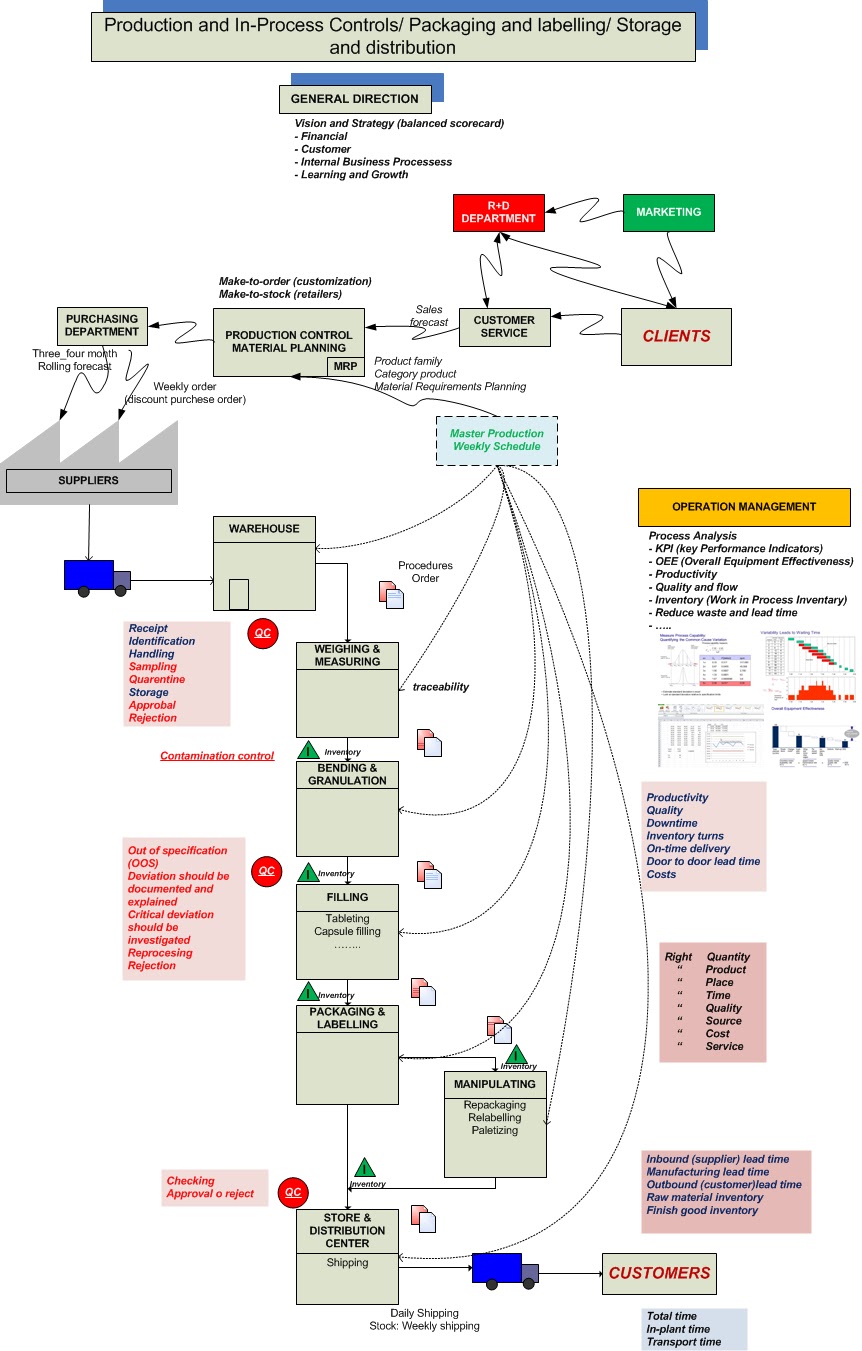
The necessity and power of Processes (Continuous
improvement)
The power of processes:
the recipes or checklists of how to do something.
Processes reduce
delegation risks and mistakes.
Processes must be written down.
After each mistake is
corrected, write a process. Apply PDCA role.
Best practices
Learning to live with the
humanity of employees—mistakes will happen!
Expect mistakes. Look for
them! Find them! Teach and correct!
For control business need
daily and weekly measurements.
• Measure: quality; on-time
delivery; customer satisfaction; efficiency; daily costs.
Engage employees in
writing processes.
Engage employees in
measuring key indicators.
Encourage and reward
self-reporting of mistakes. Mistakes WILL happen.
What is High Employee Engagement
(“HEE”)?
- Employees are emotionally engaged and personally invested in doing great work.
- Employees view their work as more than a paycheck.
- Employees are proud of where they work.
- Employees find meaning and emotional satisfaction in being part of the business.
- Employees find meaning and emotional satisfaction in the purpose of their work.
The money $$$ is not enough
- It cannot buy long-term, consistent, high employee engagement.
- Through a combination of culture, values, leadership behaviors, how is treat people—emotionally, financially—and providing an opportunity for them to be all that they can be.
- Do you care about them as people or are they just a means to your end: $$?
- Behaviors count.
HEE—HPO
(High Performance Organization)----successful business
Common characteristics of HEE/ high performance
companies
- Employees have a sense of “ownership.”
- Employees have training and educational opportunities to grow.
- “Promotion from within” policies.
- Constant communication about values and standards.
- Highly accountable, “family” environment.
- Consistent and fair reward and promotion policies.
Creating a
great place to work apply Culture of “7 E's”: Expectations, Excellence, Employees, Empowerment,
Education, Earnings and Execution.
Source: Edward D. Hess. University of Virginia